Uterine fibroids are noncancerous growths of the uterus that often appear during childbearing years. Also called fibromas, leiomyomas or myomas, uterine fibroids aren't associated with an increased risk of uterine cancer and almost never develop into cancer.
As many as three out of four women have uterine fibroids, but most are unaware of them because they often cause no symptoms. The doctor may discover them incidentally during a pelvic exam or prenatal ultrasound.
In general, uterine fibroids cause no problems and seldom require treatment. Medical therapy and surgical procedures can shrink or remove fibroids if there is no discomfort or troublesome symptoms. Rarely, fibroids can require emergency treatment if they cause sudden, sharp pelvic pain.
Signs and Symptoms
Uterine fibroids are often asymptomatic. Rapidly enlarging tumors may cause pelvic pain. They are a common cause of heavy or irregular menstrual bleeding. In women of child bearing age, these tumors may produce infertility. Excessive blood loss can lead to signs of anemia, including fatigue, shortness of breath, rapid heart rate and pallor.
Causes
Uterine fibroids develop from the smooth muscular tissue of the uterus (myometrium). A single cell reproduces repeatedly, eventually creating a pale, firm, rubbery mass distinct from neighboring tissue.
Fibroids range in size from seedlings, undetectable by the human eye, to bulky masses that can distort and enlarge the uterus. They can be single or multiple, in extreme cases expanding the uterus so much that it reaches the rib cage.
Doctors don't know the cause of uterine fibroids, but research and clinical experience point to several factors:
- Genetic alterations. Many fibroids contain alterations in genes that code for uterine muscle cells.
- Hormones. Estrogen and progesterone, two hormones that stimulate development of the uterine lining in preparation for pregnancy, appear to promote the growth of fibroids. Fibroids contain more estrogen and estrogen receptors than do normal uterine muscle cells.
- Extracellular matrix (ECM). This material makes cells stick together. ECM is increased in fibroids and makes them fibrous. ECM also stores growth factors and causes biologic changes in the cells themselves.
- Other chemicals. Substances that help the body maintain tissues, such as insulin-like growth factor, may affect fibroid growth.
Diagnosis
Uterine fibroids are frequently found incidentally during a routine pelvic exam. The doctor may feel irregularities in the shape of the uterus through the abdomen, suggesting the presence of fibroids.
Ultrasound
If confirmation is needed, the doctor may obtain an ultrasound — a painless exam that uses sound waves to obtain a picture of the uterus — to confirm the diagnosis and to map and measure fibroids. A doctor or technician moves the ultrasound device (transducer) over the abdomen (transabdominal) or places it inside the vagina (transvaginal) to obtain images of the uterus.
Transvaginal ultrasound provides more detail because the probe is closer to the uterus. Transabdominal ultrasound visualizes a larger anatomic area. Sometimes, fibroids are discovered during an ultrasound conducted for a different purpose, such as during a prenatal ultrasound.
Other imaging tests
If traditional ultrasound doesn't provide enough information, the doctor may order other imaging studies, such as:
- Hysterosonography. This ultrasound variation uses sterile saline to expand the uterine cavity, making it easier to obtain interior images of the uterus. This test may be useful if there is heavy menstrual bleeding despite normal results from traditional ultrasound.
- Hysterosalpingography. This technique uses a dye to highlight the uterine cavity and fallopian tubes on X-ray images. The doctor may recommend it if infertility is a concern. In addition to revealing fibroids, it can help the doctor determine if the fallopian tubes are open.
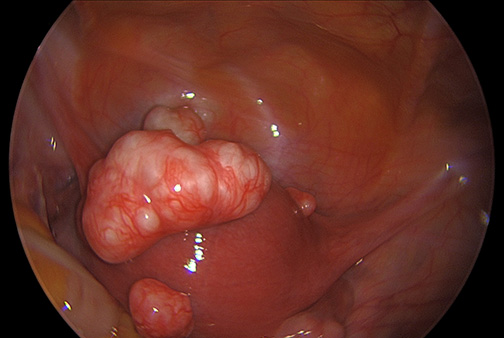
- Hysteroscopy. The doctor inserts a small, lighted telescope called a hysteroscope through the cervix into the uterus. The tube releases a gas or liquid to expand the uterus, allowing the doctor to examine the walls of the uterus and the openings of the fallopian tubes. A hysteroscopy can be performed in the doctor’s office.
Imaging techniques that may occasionally be necessary include computerized tomography (CT) and magnetic resonance imaging (MRI).
Other tests
If the client is experiencing abnormal vaginal bleeding, the doctor may want to conduct other tests to investigate potential causes. He or she may order a complete blood count (CBC) to determine if there is iron deficiency anemia because of chronic blood loss. The doctor may also order blood tests to rule out bleeding disorders and to determine the levels of reproductive hormones produced by the ovaries.
Risk Factors
Race: While uterine fibroids can develop in individuals of reproductive age regardless of their race, Black individuals exhibit a higher likelihood of experiencing fibroids compared to other racial groups. Furthermore, Black individuals tend to develop fibroids at a younger age than their white counterparts, and they may also experience a higher number of larger fibroids, often accompanied by more severe symptoms.
Family History: If your mother or sister has a history of fibroids, your risk of developing them is increased.
Other Factors: Commencing menstruation before the age of 10, obesity, low on vitamin D, a diet rich in red meat and less in green vegetables, fruits, and dairy products, as well as alcohol consumption (including beer), all appear to increase the likelihood of developing uterine fibroids.
Pathogenesis / Pathophysiology
Fibroids are monoclonal tumors, approximately 40 to 50% show karyotypically detectable chromosomal abnormalities.
Their growth is strongly dependent on estrogen and progesterone. Although both estrogen and progesterone are usually regarded as growth promoting, they will also cause growth restriction in some circumstances. Paradoxically fibroids will rarely grow during pregnancy despite very high steroid hormone levels and pregnancy appears to exert a certain protective effect.
It is believed that estrogen and progesterone have both mitogenic effect on leiomyoma cells and also act by influencing (directly and indirectly) a large number of growth factors, cytokines and apoptotic factors as well as other hormones. Furthermore the actions of estrogen and progesterone are modulated by the cross-talk between estrogen, progesterone and prolactin signaling which controls the expression of the respective nuclear receptors. It is believed that estrogen is growth promoting by up-regulating IGF-1, EGFR, TGF-beta1, TGF-beta3 and PDGF, promotes aberrant survival of leiomyoma cells by down-regulating p53, increasing expression of the anti-apoptotic factor PCP4 and antagonizing PPAR-gamma signaling. Progesterone is thought to promote the growth of leiomyoma through up-regulating EGF, TGF-beta1 and TGF-beta3, and the survival through up-regulating Bcl-2 expression and down-regulating TNF-alpha. Progesterone is believed to counteract growth by downregulating IGF-1.
Whereas in premenopausal fibroids the ER-beta, ER-alpha and progesterone receptors are found overexpressed, in the rare postmenopausal fibroids only ER-beta was found significantly overexpressed. Most studies found that polymorphisms in ER and PR gene encodings are not correlated with incidence of fibroids in Caucasian populations however a special ER-alpha genotype was found correlated with incidence and size of fibroids. The higher prevalence of this genotype in black women may also explain the high incidence of fibroids in this group.
Aromatase and 17beta-hydroxysteroid dehydrogenase are aberrantly expressed in fibroids, indicating that fibroids can convert circulating androstenedione into estradiol. Similar mechanism of action has been elucidated in endometriosis and other endometrial diseases. Aromatase inhibitors are currently considered for treatment, at certain doses they would completely inhibit estrogen production in the fibroid while not largely affecting ovarian production of estrogen (and thus systemic levels of it). Aromatase overexpression is particularly pronounced in Afro-American women.
Genetic and hereditary causes are being considered and several epidemiologic findings indicate considerable genetic influence especially for early onset cases. First degree relatives have a 2.5-fold risk, and nearly 6-fold risk when considering early onset cases. Monozygotic twins have double concordance rate for hysterectomy compared to dizygotic twins.
Like keloids, fibroids have dysregulated production of extracellular matrix. Recent studies suggest that this production may represent an abnormal response to ischemic and mechanical tissue stress. Several factors indicate significant involvement of extracellular signaling pathways such as ERK1 and ERK2, which in fibroids are prominently influenced by hormones. Paradoxically and unlike most other conditions involving significant fibrosis the Cyr61 gene has been found downregulated in fibroids.
Cyr61 is also known for its role as tumor suppressing factor and in angiogenesis. Hence fibroids are one of the very few tumors with reduced vascular density.
Prevention
There is no known treatment that prevents uterine fibroids. But getting regular exercise may help. According to one study, the more exercise women have, the less likely they are to get uterine fibroids.
Preventing fibroids from coming back after treatment
It is common for fibroids to grow back after treatment. The only treatment that absolutely prevents regrowth of fibroids is removal of the entire uterus, called hysterectomy. After hysterectomy, you cannot get pregnant. While many women report an improved quality of life after hysterectomy, there are also possible long-term side effects to consider.
Nursing Interventions
- Educate the patient about uterine fibroids. Nurses can provide patients with information about the condition, including the causes, symptoms, and treatment options. They can also help patients to understand their individual risk factors and how to manage their symptoms.
- Assess the patient's symptoms and needs. Nurses can work with patients to identify their individual symptoms and needs. This information can be used to develop a personalized treatment plan.
- Provide support and counseling. Nurses can provide emotional support and counseling to patients with uterine fibroids. They can help patients to cope with the stress of their condition and make decisions about their care.
- Monitor the patient's condition. Nurses can monitor patients with uterine fibroids for changes in their symptoms and overall health. They can also monitor the effectiveness of treatment and make adjustments as needed.
- Provide symptom management. Nurses can provide patients with medications and other treatments to relieve their symptoms. They can also teach patients about self-management strategies, such as pain management and heat therapy.
Coordinate care with other healthcare providers. Nurses can work with other healthcare providers, such as doctors and specialists, to coordinate the patient's care. This helps to ensure that the patient receives the best possible care.
Here are some additional nursing interventions that can be used to manage specific symptoms of uterine fibroids:
- Heavy menstrual bleeding: Nurses can teach patients about ways to manage heavy menstrual bleeding, such as using iron supplements and menstrual cramps.
- Pelvic pain: Nurses can teach patients about pain management strategies, such as heat therapy and relaxation techniques.
- Pressure on the bladder or bowels: Nurses can teach patients about ways to manage pressure on the bladder or bowels, such as avoiding caffeine and spicy foods.
Nurses can also play a role in preventing uterine fibroids. They can educate patients about the risk factors for uterine fibroids and how to reduce their risk. Nurses can also encourage patients to see their doctor for regular checkups, so that any fibroids can be detected and treated early.
Treatment
There's no single best approach to uterine fibroid treatment. Many treatment options exist. In most cases, the best action to take after discovering fibroids is simply to be aware they are there.
Watchful waiting
Women with uterine fibroids have no signs or symptoms. In this case, watchful waiting (expectant management) could be the best course. Fibroids aren't cancerous. They rarely interfere with pregnancy. They usually grow slowly and tend to shrink after menopause when levels of reproductive hormones drop. This is the best treatment option for a large majority of women with uterine fibroids.
Medications
Medications for uterine fibroids target hormones that regulate menstrual cycle, treating symptoms such as heavy menstrual bleeding and pelvic pressure. They don’t eliminate fibroids, but may shrink them. Medications include:
- Gonadotropin-releasing hormone (Gn-RH) agonists. To trigger a new menstrual cycle, a control center in the brain called the hypothalamus manufactures gonadotropin-releasing hormone (Gn-RH). The substance travels to the pituitary gland, a tiny gland also located at the base of the brain, and sets in motion events that stimulate the ovaries to produce estrogen and progesterone.
Medications called Gn-RH agonists (Lupron, Synarel, others) act at the same sites that Gn-RH does. But when taken as therapy, a Gn-RH agonist produces the opposite effect to that of the natural hormone. Estrogen and progesterone levels fall, menstruation stops, fibroids shrink and anemia often improves.
- Androgens. The ovaries and the adrenal glands, located above the kidneys, produce androgens, the so-called male hormones. Given as medical therapy, androgens can relieve fibroid symptoms.
Danazol, a synthetic drug similar to testosterone, has been shown to shrink fibroid tumors, reduce uterine size, stop menstruation and correct anemia. However, occasional unpleasant side effects such as weight gain, dysphoria (feeling depressed, anxious or uneasy), acne, headaches, unwanted hair growth and a deeper voice, make many women reluctant to take this drug.
Other medications
Oral contraceptives or progestins can help control menstrual bleeding, but they don't reduce fibroid size. Nonsteroidal anti-inflammatory drugs (NSAIDs), which are not hormonal medications, are effective for heavy vaginal bleeding unrelated to fibroids, but they don't reduce bleeding caused by fibroids.
Hysterectomy
This operation — the removal of the uterus — remains the only proven permanent solution for uterine fibroids. But hysterectomy is major surgery. It ends the ability to bear children, and if elected to have ovaries removed also, it brings on menopause and the question of whether to take hormone replacement therapy.
Myomectomy
In this surgical procedure, the surgeon removes the fibroids, leaving the uterus in place. If a patient wants to bear children, this is the ideal option. With myomectomy, as opposed to a hysterectomy, there is a risk of fibroid recurrence. There are several ways a myomectomy can be done:
- Abdominal myomectomy. If having multiple fibroids, very large or very deep fibroids, the doctor may use an open abdominal surgical procedure to remove the fibroids.
- Laparoscopic myomectomy. If the fibroids are small and few in number, the client and the doctor may opt for a laparoscopic procedure, which uses slender instruments inserted through small incisions in the abdomen to remove the fibroids from the uterus. The doctor views the abdominal area on a remote monitor via a small camera attached to one of the instruments.
- Hysteroscopic myomectomy. This procedure may be an option if the fibroids are contained inside the uterus (submucosal). A long, slender scope (hysteroscope) is passed through the vagina and cervix and into the uterus. The doctor can see and remove the fibroids through the scope. This procedure is best performed by a doctor experienced in this technique.
Variations of myomectomy — in which uterine fibroids are destroyed without actually removing them — include:
- Myolysis. In this laparoscopic procedure, an electric current destroys the fibroids and shrinks the blood vessels that feed them.
- Cryomyolysis. In a procedure similar to myolysis, cryomyolysis uses liquid nitrogen to freeze the fibroids. The safety, effectiveness and associated risk of fibroid recurrence of myolysis and cryomyolysis have yet to be determined.
- Endometrial ablation. This treatment, performed with a hysteroscope, uses heat to destroy the lining of the uterus, either ending menstruation or reducing the menstrual flow. Endometrial ablation is effective in stopping abnormal bleeding, but doesn't affect fibroids outside the interior lining of the uterus.
Uterine artery embolization
Small particles injected into the arteries supplying the uterus cut off blood flow to fibroids, causing them to shrink. This technique is proving effective in shrinking fibroids and relieving the symptoms they can cause. Advantages over surgery include:
· No incision
· Shorter recovery time
Complications may occur if the blood supply to the ovaries or other organs is compromised.
Focused ultrasound surgery
MRI-guided focused ultrasound surgery (FUS), approved by the Food and Drug Administration in October 2004, is a newer treatment option for women with fibroids. Unlike other fibroid treatment options, FUS is noninvasive and preserves the uterus.
This procedure is performed while the client is inside of a specially crafted MRI scanner that allows doctors to visualize the anatomy, and then locate and destroy (ablate) fibroids inside the uterus without making an incision. Focused high-frequency, high-energy sound waves are used to target and destroy the fibroids. A single treatment session is done in an on- and off-again fashion, sometimes spanning several hours. Initial results with this technology are promising, but its long-term effectiveness is not yet known.
Complications
Although uterine fibroids usually aren't dangerous, they can cause discomfort and may lead to complications such as anemia from heavy blood loss. In rare instances, fibroid tumors can grow out of the uterus on a stalk-like projection. If the fibroid twists on this stalk, a sudden, sharp, severe pain in the lower abdomen may develop. If so, seeking medical care is strongly recommended. Surgery may be needed.
Pregnancy and fibroids
Because uterine fibroids typically develop during the childbearing years, women with fibroids are often concerned about their chances of a successful pregnancy.
Fibroids usually don't interfere with conception and pregnancy, but they can occasionally affect fertility. They may distort or block the fallopian tubes, or interfere with the passage of sperm from the cervix to the fallopian tubes. Submucosal fibroids may prevent implantation and growth of an embryo.
Research indicates that pregnant women with fibroids are at slightly increased risk of miscarriage, premature labor and delivery, abnormal fetal position, and separation of the placenta from the uterine wall. But not all studies confirm these associations. Furthermore, complications vary based on the number, size and location of fibroids. Multiple fibroids and large submucosal fibroids that distort the uterine cavity are the type most likely to cause problems. A more common complication of fibroids in pregnancy is localized pain, typically between the first and second trimesters. This is usually easily treated with pain relievers.
In most cases, fibroids don't interfere with pregnancy and treatment isn't necessary. It was once believed that fibroids grew faster during pregnancy, but multiple studies suggest otherwise. Most fibroids remain stable in size, although some increase or decrease slightly, usually in the first trimester.
If there are fibroids and the patient experienced repeated pregnancy losses, the doctor may recommend removing one or more fibroids to improve the chances of carrying a baby to term, especially if no other causes of miscarriage can be found and the fibroids distort the shape of the uterine cavity.
Doctors usually don't remove fibroids in conjunction with a Caesarean section because of the high risk of excessive bleeding.