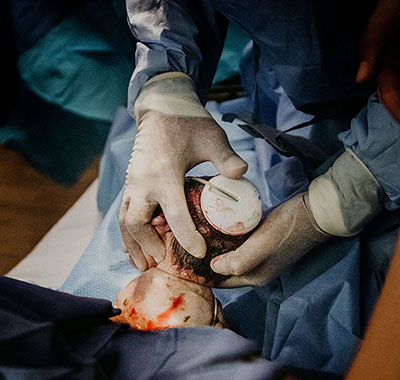
Vacuum Extraction or VE is a medical procedure that involves removal of a fetus from the uterus or vagina via a suction device such as a vacuum extractor or a ventouse.
Prerequisites for Vacuum Extraction
The indications for vacuum extraction (VE) operations are the same as assisted delivery operations performed with forceps. The prerequisites for all types of instrumental delivery are discussed below.
Informed consent
Informed consent is required for any surgical procedure, including an instrumental delivery. Informed consent is best considered as a process and not simply a signed form. Consent for a surgical procedure requires an explanation of the need for the operation, a discussion of risks and benefits, and a presentation of alternative modes of treatment. The patient must also be given the opportunity to ask questions. This consent process may strike the clinician as time consuming and unrealistic for an instrumental delivery, especially in the face of urgency. However, the potential for maternal or fetal injury and thus the medical and legal risks from an assisted delivery are substantially greater than those associated with many other surgical procedures wherein consent is routinely obtained.
A bedside consent process can be abbreviated, especially in cases of presumed fetal jeopardy (non-reassuring fetal monitoring). In all cases, excluding extreme emergencies, sufficient time is available to briefly describe the proposed operation to the mother, review the indications, and state the limits of effort intended.
Routinely discussing possible obstetric interventions with families at an earlier time during the pregnancy is important because of the controversy concerning bedside consents in acute situations. When antepartum discussions have occurred and the need for an instrumental delivery procedure is later presented during labor, both the patient and family will already have a general idea about the procedure and its potential risks and benefits.
Prepared physician
The clinician must have knowledge of the instrument chosen, VE indications, and proven techniques. The decision to perform instrumentation should follow an analysis of the course in labor, a pelvic examination, and determination of fetal position and station and consideration of the fetal/pelvic relationship. Most importantly, the accoucheur must be prepared to reconsider or abandon any operation that proves difficult.
Prepared patient
The initial requirement is consent. Thereafter and prior to an extraction attempt, the patient should have ruptured membranes; empty bladder by Credé, catheterization, or spontaneous voiding; full cervix dilation; an engaged fetal head; and no suspicion of fetopelvic disproportion. If the fetal position or the station of the presenting part is uncertain, a transperineal or transvaginal real time ultrasonographic examination can be performed before attempting the operation. Ultrasonography can also be used to judge the appropriateness of the vacuum cup application. These procedures are easily performed at the bedside. Position is readily identified by noting the fetal orbits and identifying characteristic intracranial anatomy (falx, posterior fossa, etc). Station can be estimated by translabial scanning and requires more experience.
Acceptable analgesia/anesthesia
Some outlet operative VE deliveries can be conducted without anesthesia or analgesia. However, parturients do find operative vaginal procedures painful. Usually, either a regional anesthetic (eg, pudendal block) or, more frequently (and more effectively), a conduction anesthetic (eg, epidural, spinal, saddle block) is required.
Indications for Vacuum Extraction
- Prolonged pushing in the second stage of labor or maternal exhaustion
- Fetal emergency in the second stage of labor, generally indicated by changes in the fetal heart rate
- Maternal illness where "bearing down" or pushing efforts would be risky (e.g. cardiac conditions, blood pressure)
Contraindications to Vacuum Extraction
Vacuum operation is contraindicated in the following instances:
- Operator inexperience
- Inability to achieve a correct application
- An inadequate trial of labor
- Lack of a standard indication
- Uncertainty concerning fetal position and station
- Suspicion of fetopelvic disproportion
- Fetal malpositioning (eg, breech, face, brow)
- Known or suspected fetal coagulation defects
Relative contraindications are as follows:
- Prematurity (fetus <36 wk gestation): Vacuum extraction (VE) applications are not recommended at less than 36 weeks’ gestation. Data are limited and the magnitude of risk is not known with certainty. The physics of scalp entrapment by the cup mechanism and the known fragility of intracranial vascular structures in the premature infant mitigate against such applications unless special circumstances exist.
- Prior scalp sampling: Scalp sampling is now an uncommon procedure. A prior sampling is not normally a problem in a VE operation. In very unusual circumstances, fetal bleeding has occurred with VE in this setting but the risk is remote at best. Prior application of a scalp electrode does not preclude VE use.
- Prior failed forceps: Prior failed forceps is usually a contraindication to any VE effort. As a practical matter, the forceps can generate more traction force than the VE. Thus, to follow a forceps effort with a VE trial is not inherently reasonable unless circumstances have prevented the successful application of a forceps before traction has been attempted. An example might be the inability to achieve (or properly verify) an appropriate forceps application after blade insertion. Such cases must be quite unusual and should prompt immediate caution as heavy cranial molding and advanced caput (both harbingers of disproportion) are likely to be present.
- Overlapping cranial bones, heavy caput: Overlap of cranial bones, the inability to palpate the standard landmarks, and cranial edema (caput/cephalohematoma) are suggestive of at least relative disproportion. When these findings are combined with poor progress, the clinician may find that the presenting part is substantially higher than initially anticipated. True disproportion may be present, precluding any vaginal trial. As previously noted, the bedside evaluation of such cases when real time ultrasonographic scanning can be of particular assistance.
- Known or suspected fetal macrosomia: A large infant (estimated fetal weight >4,500 g in a nondiabetic) can only be considered as a relative contraindication to instrumental delivery. This is primarily because the methods of estimation of fetal bulk are so limited and inaccurate. While ultrasonographic scanning is often used for weight estimates at or near term, in most cases, such estimates are considered as +/- 6-12%, with only 40-75% of these estimates falling within this range. These ultrasonographic data should not be used as the sole means for the estimation of fetal bulk or for making obstetric management decisions. This information is only useful to the clinician when combined with other information that dynamically evaluates the fetopelvic relationship (eg, Muller-Hillis maneuver, overlap of cranial bones, course in labor, pelvimetry).
Technique
The woman is placed in the lithotomy position and assists throughout the process by pushing. A suction cup is placed onto the head of the baby and the suction draws the skin from the scalp into the cup. Proper placement is critical to keep the head flexed, thus the cup is placed on the flexion point, about 3 cm anterior from the occipital (posterior) fontanelle. Ventouse devices have handles to allow for traction. When the head is born, the device is detached, allowing the woman to complete the delivery of her child.
Generally the cervix has to be fully dilated, the head engaged, and the head position known to allow for a proper use of the ventouse. If the ventouse attempt fails it may be necessary to deliver the infant by caesarean section.
Birth Injuries
Neonatal Injury
No type of assisted delivery is entirely free of risk for both mother and infant. The issues are the magnitude of the risk and the alternatives available for delivery once the natural processes fail. The reported incidence of fetal death or severe fetal injury from vacuum extraction (VE) is low, ranging from 0.1-3 cases per 1,000 extraction procedures. Clinically diagnosed scalp injuries occur largely because of the physics of vacuum extraction. As the vacuum force is applied, the extractor draws the fetal scalp into the body of the cup. This produces the characteristic mound of scalp tissue and edema, the chignon, that may be identified after an extraction. Traction also tensions the scalp against its attachments to the fetal skull, drawing it in the direction of the cup. These effects predispose to bleeding within the substance of the scalp.
The 2 major types of scalp injury are the common, but clinically unimportant, cephalohematomas and the relatively rare, but potentially life threatening, subgaleal hemorrhages. Scalp bruising or lacerations and retinal hemorrhages are additional, usually insignificant fetal risks of extraction procedures.
When either radiographic or ultrasonic studies of the CNS are routinely performed on newborns who were delivered spontaneously or by instrumental assistance, minor trauma to the scalp, linear occult cranial fractures and subarachnoid bleeding are discovered more frequently than their clinical signs and symptoms suggest. A small series by Whitby using MRI studies in term asymptomatic newborns reports similar findings.
These observations and the difficulties in clinically distinguishing cephalohematomas from edema or in diagnosing small subgaleal bleeds indicate that investigations based solely on clinical presentation and physical examinations miss many asymptomatic neonates with some degree of birth-related trauma. The data also indicate that the large proportion of these asymptomatic events are not of clinical importance. Drawing from the Towner data, the major intervening risk for these occult injuries is probably the combination of labor and the cranial distortion normal in vaginal delivery.
Subgaleal and subaponeurotic hemorrhage
The most feared complication of VE is hemorrhage in the subgaleal (SG) or subaponeurotic space from rupture of the emissary veins. This condition is potentially life threatening, with a mortality rate reported as high as 20%. Approximately half of all SG hemorrhages are related to VE. Most of the rest are associated with forceps operations. Less commonly, SG bleeds follow spontaneous deliveries. The reported incidence of clinically identified or symptomatic SG hemorrhages ranges from 6-50 per 1,000 VE operations. These rates almost certainly are overestimates and do not reflect the rates of injury in modern practice when soft-cup extractors are used and strict protocols for application and the limitation of effort are followed.
SG bleeding was not observed in the large number of cases included in several major clinical reports in recent years. No SG cases were reported in the large vacuum extraction meta-analysis by Johanson. Also, In the 2007 report by Simonson and coworkers, of the 913 VE procedures, no instances were reported in which the neonates were routinely screened postdelivery by cranial radiography. Further, the French study by Baume and coworkers involving 845 VE operations also did not report any clinically identified cases of SG hemorrhage. These data document the rarity of such clinically significant severe scalp injuries. These results also emphasize that when strict technical guidelines for the performance of VE operations are used, the risk of symptomatic SG hemorrhage while never zero is, nonetheless, quite small.
According to some authors, SG bleeding is most likely when excessive force, multiple pop-offs, prolonged cup application times or serial instrumentation with VE and forceps has occurred. However, recall that serious cases of SG bleeding have followed outwardly uneventful extractions. Thus, even adherence to protocol may not avoid all cases. Because of the small but significant risk of SG bleeding, the attending pediatric personnel should be informed whenever a VE operation has occurred, regardless of the immediate condition of the neonate. SG hemorrhages are dangerous because the clinical signs and symptoms may not be clinically apparent until some hours postpartum. Thus, serial observation is required.
Scalp bruising and lacerations
Ecchymoses and, uncommonly, scalp slough or lacerations can follow VE. Most of these injuries occur when the recommended 30-minute (some authors favor 20 minutes) limit to total cup application is exceeded or efforts are made at cup rotation.
The ventouse is not a rotating instrument. Attempts at cup rotation simply foster cup displacement, loss of station, or scalp injury. Under traction, the fetal head rotates automatically as descent occurs. If the clinician feels an obligation to assist or hasten this process, then manual rotation of the head (not the cup) can accompany the extraction. Normally, this is not required and efforts at rotation should never be forced.
Maternal Injury
Vacuum extraction has a low rate of maternal injury in comparison with forceps operations or cesarean delivery. However, maternal injuries do occur. Such trauma cannot be disregarded in evaluating the risk of the procedure.
Lacerations
Maternal perineal lacerations are common complications of all operative vaginal deliveries. Many tears are associated with episiotomy. Depending on series reflecting obstetric routines of perineal management and instrument use, the incidence of severe perineal lacerations (ie, third- and fourth-degree lacerations) during VE procedures ranges from 5-30%. Women who sustain vaginal lacerations in a previous delivery are at a significantly greater risk for a repeat laceration in subsequent deliveries. Women at greatest risk are those who experienced a laceration in the first delivery followed by another delivery combining both an instrumental delivery and an episiotomy. Delivery technique, skill, fetal bulk, prior scars, and instrument choice are important factors in perineal injury.
One area in which vacuum extractor has a clear advantage over forceps is the incidence of perineal trauma. Forceps operations are more likely to result in anal sphincter injury trauma than vacuum extractions. Episiotomy is an important risk factor. Electively incising the perineum predisposes to perineal lacerations and rectal injuries by direct extension. Some authors favor the selective performance of episiotomy and only if maternal soft tissues impede the delivery process. In Europe, when an episiotomy is required, mediolateral (ML) incisions are preferred. ML episiotomies are less likely than median episiotomies (ME) to extend into the rectal sphincter or mucosa. However, the ML is harder to repair, is more likely to result in distortion of the perineum, and results in more pain in the puerperium.
Episiotomy should not be routine and careful technique can often avoid tears, especially in multiparas. Best practice concerning whether to perform an episiotomy during an instrumental delivery, the type to use, and the timing is yet to be established. Some authors say that episiotomy should be avoided when possible and, if required, the type depends on maternal anatomy.
Stress, urinary, and anal incontinence
Genetic predisposition, dystocia in labor, vaginal delivery, obstetric lacerations, multiparity, and probably the mode of delivery combine to result in both reversible and permanent injuries to connective tissues of the maternal pelvis. Injury to these support structures and to the rectum constitutes important and perhaps unavoidable risks of both labor and instrumental delivery.
The female pelvic viscera are suspended from above and supported from below. The intactness of the various support structures depends upon the integrity of their muscular, fascial, and neurologic constituents.
The upper suspensory structures are a complex of pseudoligamentous structures loosely termed pelvic ligaments. This connective tissue accompanies vascular structures into the pelvis to surround the cervix. The lower supports for the uterus are a musculofascial complex including the urogenital and pelvic diaphragms. The pelvic diaphragm principally consists of the levator ani muscle. The urogenital diaphragm is a complex of small muscles and accompanying connective tissue that extends from the central perineal body radially to attach to various bony and ligamentous sites in the pelvis.
Both labor and the process of passing the fetal body through the birth canal distort and injure these and other pelvic tissues. During parturition, pelvic ligaments and muscles are simply torn or otherwise disrupted and accompanying nerves are traumatized. Various spontaneous lacerations or episiotomy extensions account for additional injuries, especially to the rectal sphincter.
The issue is not whether vaginal delivery results in injuries to pelvic soft tissues. The question is the degree of the injury and the extent to which spontaneous postpartum healing or specific muscle strengthening exercises performed in the puerperium may ameliorate this damage. In terms of instrumental delivery, techniques that either reduce or avoid injury to pelvic supports and to the rectum are under study. Long-term, follow-up studies controlling for prepartum pelvic support status (eg, preexisting rectal dysfunction, urinary incontinence) as well as length of labor, type of anesthesia, clinically observed perineal trauma, and delivery method are required before changes in current practice can be confidently recommended.
Nursing Interventions
Vacuum Extraction can only be performed by trained practitioners. What nurses can do and should do are the following:
- Before the procedure:
- Assess the mother's and fetus' condition.
- Explain the procedure to the mother and answer any questions she may have.
- Obtain informed consent.
- Ensure that the necessary equipment is available and in working order.
- During the procedure:
- Assist the healthcare provider with positioning the mother and applying the vacuum cup.
- Monitor the mother's and fetus' vital signs.
- Provide support and comfort to the mother.
- Be prepared to assist with any complications that may arise.
- After the procedure:
- Monitor the mother's and fetus' vital signs.
- Assess the mother's perineum for any injuries.
- Provide pain relief as needed.
- Assist the mother with breastfeeding.
- Educate the mother on postpartum care.
The nurse should also watch out for complications. Be prepared to assist with any complications that may arise, such as fetal distress, hemorrhage, or perineal lacerations.