Tuberculosis (abbreviated as TB for tubercle bacillus or Tuberculosis) is a common and deadly infectious disease caused by mycobacteria, mainly Mycobacterium tuberculosis. Tuberculosis most commonly attacks the lungs (as pulmonary TB) but can also affect the central nervous system, the lymphatic system, the circulatory system, the genitourinary system, bones, joints, the digestive system, and even the skin. Other mycobacteria such as Mycobacterium bovis, Mycobacterium africanum, Mycobacterium canetti, and Mycobacterium microti can also cause tuberculosis, but these species do not usually infect healthy adults.
The bacillus causing tuberculosis, Mycobacterium tuberculosis, was identified and described on March 24, 1882 by Robert Koch. He received the Nobel Prize in physiology or medicine in 1905 for this discovery. Koch did not believe that bovine (cattle) and human tuberculosis were similar, which delayed the recognition of infected milk as a source of infection. Later, this source was eliminated by the pasteurization process. Koch announced a glycerine extract of the tubercle bacilli as a "remedy" for tuberculosis in 1890, calling it 'tuberculin'. It was not effective, but was later adapted as a test for pre-symptomatic tuberculosis.
Over one-third of the world's population has been exposed to the TB bacterium, and new infections occur at a rate of one per second. Not everyone infected develops the full-blown disease, so asymptomatic, latent TB infection is most common. However, one in ten latent infections will progress to active TB disease, which, if left untreated, kills more than half of its victims.
TB may developed resistance to antibiotics if mismanaged, this results to a more formidable form of TB.
Other Names
TB, Consumption (old name), phthisis (Greek for consumption), phthisis pulmonalis, Scrofula (skin TB), wasting disease, white plague, king’s evil (old name for skin TB), Pott’s disease (extrapulmonary TB).
Signs and Symptoms
When the disease becomes active, 75% of the cases are pulmonary TB. Symptoms include chest pain, coughing up blood, and a productive, prolonged cough for more than three weeks. Systemic symptoms include fever, chills, night sweats, appetite loss, weight loss, pallor, and often a tendency to fatigue very easily.
In the other 25% of active cases, the infection moves from the lungs, causing other kinds of TB more common in immunosuppressed persons and young children. Extrapulmonary infection sites include the pleura, the central nervous system in meningitis, the lymphatic system in scrofula of the neck, the genitourinary system in urogenital tuberculosis, and bones and joints in Pott's disease of the spine. An especially serious form is disseminated TB, more commonly known as miliary tuberculosis. Although extrapulmonary TB is not contagious, it may co-exist with pulmonary TB, which is contagious.
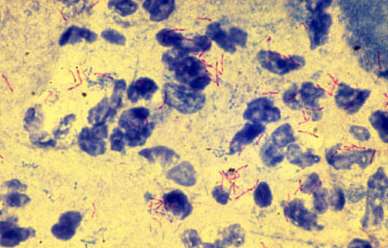
Causative Agent
Mycobacterium tuberculosis
Mode of Transmission
TB is transmitted mainly by inhalation of infectious droplets produced by persons with pulmonary or laryngeal tuberculosis during coughing, laughing, shouting or sneezing.
Invasion may occur through mucous membranes or damaged skin.
Extrapulmonary tuberculosis, other than laryngeal infection, is generally not communicable. Urine is infectious in cases of renal tuberculosis. Bovine tuberculosis results mainly from ingestion of unpasteurized milk and dairy products. Aerosol transmission has been reported among abattoir workers.
Diagnosis
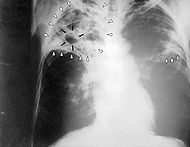
Tuberculosis can be a difficult disease to diagnose, due mainly to the difficulty in culturing this slow-growing organism in the laboratory. A complete medical evaluation for TB must include a medical history, a chest X-ray, and a physical examination. Tuberculosis radiology is used in the diagnosis of TB. It may also include a tuberculin skin test using Mantoux procedure, a serological test, microbiological smears and cultures. The interpretation of the tuberculin skin test depends upon the person's risk factors for infection and progression to TB disease, such as exposure to other cases of TB or immunosuppression.
Currently, latent infection is diagnosed in a non-immunized person by a tuberculin skin test, which yields a delayed hypersensitivity type response to purified protein derivatives of M. tuberculosis. Those immunized for TB or with past-cleared infection will respond with delayed hypersensitivity parallel to those currently in a state of infection and thus the test must be used with caution, particularly with regard to persons from countries where TB immunization is common. New TB tests are being developed that offer the hope of cheap, fast and more accurate TB testing. These use polymerase chain reaction detection of bacterial DNA and antibody assays to detect the release of interferon gamma in response to mycobacteria. Rapid and inexpensive diagnosis will be particularly valuable in the developing world.
Incubation Period
The time from the receipt of infection to the development of a positive tuberculosis test ranges from three to six weeks, and thereafter. The development of the disease depends upon the closeness of contact, extent of the disease and sputum positivity of the source case (dose of infection) and host–parasite relationship. Thus, the incubation period may be weeks, months or years.
Pathogenesis / Pathophysiology
M. tuberculosis primarily is spread as an airborne aerosol from infected to noninfected individuals. The organisms gain access to susceptible hosts through the lung. Initial TB infection usually results in a latent or dormant infection in hosts with normally functioning immune systems. M tuberculosis is a slow-growing obligate aerobe and a facultative intracellular parasite. Because of the unique ability to survive and proliferate within mononuclear phagocytes, M. tuberculosis is able to invade local lymph nodes and spread to extrapulmonary sites, usually via hematogenous routes.
Infected end organs typically have high regional oxygen tension (apices of the lungs, kidneys, bones, meninges, eye, and choroid). The principal cause of tissue destruction from M. tuberculosis infection is related to the organism's ability to incite intense host immune reactions to antigenic cell wall proteins. TB is a multisystemic disease with myriad presentations and manifestations. Mycobacteria are highly antigenic, and promote a vigorous, nonspecific immune response. Their antigenicity is due to multiple cell wall constituents, including glycoproteins, phospholipids, and wax D, which activate Langerhans cells, lymphocytes, and polymorphonuclear leukocytes.
Mycobacterium bovis is a key constituent of Freund's adjuvant, frequently used in basic immunology research to stimulate the response to injected antigens. TB is also one of the great imitators for its well-known ability to masquerade as other infectious and disease processes within the human body. The hallmark of extrapulmonary TB histopathology is the caseating granuloma consisting of giant cells with central caseating necrosis. Rarely, if ever, are any TB bacilli seen.
Prevention
TB prevention and control takes two parallel approaches. In the first, people with TB and their contacts are identified and then treated. Identification of infections often involves testing high-risk groups for TB. In the second approach, children are vaccinated to protect them from TB. Unfortunately, no vaccine is available that provides reliable protection for adults. However, in tropical areas where the incidence of atypical mycobacteria is high, exposure to nontuberculous mycobacteria gives some protection against TB.
Many countries use BCG (Bacillus of Calmette and Guerin) vaccine as part of their TB control programs, especially for infants. The protective efficacy of BCG for preventing serious forms of TB (e.g. meningitis) in children is greater than 80%; its protective efficacy for preventing pulmonary TB in adolescents and adults is variable, ranging from 0 to 80%.
In South Africa, the country with the highest prevalence of TB, BCG is given to all children under the age of three. However, the effectiveness of BCG is lower in areas where mycobacteria are less prevalent, therefore BCG is not given to the entire population in these countries. In the USA, for example, BCG vaccine is not recommended except for people who meet specific criteria:
- Infants or children with negative skin-test result who are continually exposed to untreated or ineffectively treated patients or will be continually exposed to multidrug-resistant TB.
- Healthcare workers considered on an individual basis in settings in which high percentage of MDR-TB patients has been found, transmission of MDR-TB is likely, and TB control precautions have been implemented and not successful.
Several new vaccines to prevent TB infection are being developed. The first recombinant tuberculosis vaccine entered clinical trials in the United States in 2004, sponsored by the National Institute of Allergy and Infectious Diseases (NIAID). A 2005 study showed that a DNA TB vaccine given with conventional chemotherapy can accelerate the disappearance of bacteria as well as protect against re-infection in mice; it may take four to five years to be available in humans. A very promising TB vaccine, MVA85A, is currently in phase II trials in South Africa by a group led by Oxford University, and is based on a genetically modified vaccinia virus. Because of the limitations of current vaccines, researchers and policymakers are promoting new economic models of vaccine development including prizes, tax incentives and advance market commitments.
Nursing Interventions
Nursing interventions for TB patients are focused on:
- Preventing the spread of the infection: TB is spread through the air when an infected person coughs or sneezes. Nurses can help to prevent the spread of TB by educating patients about the infection and how to avoid transmitting it to others. Nurses can also help to identify and treat TB contacts, which are people who have been exposed to the TB bacteria.
- Promoting adherence to the treatment regimen: TB treatment can be long and complex, and it is important for patients to take their medications as prescribed to ensure a cure. Nurses can help patients to adhere to their treatment regimen by educating them about the importance of taking their medications, providing support and encouragement, and monitoring for any side effects.
- Managing symptoms and complications: TB can cause a variety of symptoms, such as cough, fever, night sweats, and weight loss. Nurses can help to manage these symptoms by providing medications and supportive care. Nurses can also monitor patients for complications of TB, such as respiratory failure and kidney failure.
Here are some specific nursing interventions that may be used for TB patients:
- Airborne isolation precautions: TB is spread through the air, so patients with active TB must be placed in airborne isolation precautions. This means that they must be placed in a single room with a negative pressure ventilation system. Visitors must wear a mask and gown when entering the patient's room.
- Sputum collection: TB is diagnosed by testing sputum samples for the TB bacteria. Nurses can help patients to collect sputum samples by providing them with sterile containers and instructions on how to collect the samples.
- Medication administration: TB is treated with a combination of antibiotics. Nurses can help patients to take their medications as prescribed by providing them with medication reminders and monitoring them for any side effects.
- Nutritional support: TB can cause weight loss and malnutrition. Nurses can help to ensure that TB patients are getting the nutrients they need by providing them with nutritional counseling and dietary supplements.
- Education and support: Nurses can provide TB patients with education about the infection, how to prevent the spread of the infection, and how to manage their symptoms. Nurses can also provide patients with support and encouragement throughout their treatment.
Treatment
The ATS and CDC have provided standard guidelines for the treatment of TB. The ultimate goal of treatment is to achieve sterilization of the TB lesion in the shortest possible time. The general rule is strict adherence to TB treatment regimens for a sufficient period of time. To prevent the emergence of resistance, the regimens for the treatment of TB always should consist of multiple drugs.
Current recommendations for the treatment of pulmonary TB include a 6-month course of INH and rifampin (rifampicin), supplemented during the first 2 months with pyrazinamide. Ethambutol (or streptomycin in children too young to be monitored for visual acuity) may need to be included in the initial regimen until the results of drug susceptibility studies are available. Drug susceptibility studies may not be required if the risk of drug resistance is not significant. Significant risk factors include residence in a community with greater than 4% primary resistance to INH, history of previous treatment with anti-TB drugs, history of exposure to a drug-resistant case, and origin in a country with a high prevalence of drug resistance. The purpose of this recommendation is to decrease the development of MDR-TB in areas where primary INH resistance is increased.
Another treatment option is a 2-month regimen of INH, rifampin, and pyrazinamide daily, followed by 4 months of INH and rifampin twice a week. Effective treatment of hilar adenopathy when the organisms are fully susceptible is a 9-month regimen of INH and rifampin daily or a 1-month regimen of INH and rifampin once a day followed by 8 months of INH and rifampin twice a week.
Since poor adherence to these regimens is a common cause of treatment failure, directly observed therapy (DOT) is recommended for treatment of TB. DOT means a health care provider or other responsible person must watch the patient ingest the medications. Intermittent regimens should be monitored by DOT for the duration of therapy because poor compliance may result in inadequate drug delivery.
Complications
Miliary disease and tubercular meningitis are the earliest and most deadly complications of primary TB. A high index of suspicion is required for prompt diagnosis and management of these conditions. Pulmonary complications of TB include the development of pleural effusions and pneumothorax. Complete obstruction of a bronchus can result if caseous material extrudes into the lumen. This can lead to atelectasis of the involved lung. Bronchiectasis, stenosis of the airways, bronchoesophageal fistula, and endobronchial disease caused by penetration through an airway wall are other catastrophes that may occur with primary TB.
Perforation of the small bowel, obstruction, enterocutaneous fistula, and the development of severe malabsorption may complicate TB of the small intestine.
Pericardial effusion can be an acute complication or can resemble chronic constrictive pericarditis.
Renal complications including hydronephrosis and autonephrectomy usually do not occur in children. Paraplegia may complicate Pott disease of the spine (ie, tubercular spondylitis).